This page is intended as a resource. Click here to read about our bispecific antibody engineering service.
Bispecifics are a class of engineered antibody and antibody-like proteins that, in contrast to ‘regular’ monospecific antibodies, combine two or more different specific antigen binding elements in a single construct. Since bispecific antibodies do not typically occur in nature, they are constructed either chemically or biologically, using techniques such as cell fusion or recombinant DNA technologies. The ability to bind two different epitopes with a single molecule offers a number of potential advantages. One approach is to use the specificity of one arm as a targeting site for individual molecules, cellular markers or organisms, such as bacteria and viruses. The other arm functions as an effector site for the recruitment of effector cells or delivery of molecular payloads to the target, such as drugs, cytokines or toxins. Alternatively, bispecifics can be used to dual target, allowing detection or binding of a target cell type with much higher specificity than monospecific antibodies.
Over the past forty years a wide variety of multi-specific antibody formats, and techniques to produce them, have been developed. This is driven, in part, by the need to be able to efficiently produce and purify multi-specific antibodies for use in the clinic, as well as to modify their functional properties. We will break these formats down into 5 main areas and discuss each in turn. It must be noted though that this only covers a sub-section of the many varied bi, tri and tetra-specific antibody formats that have been developed in one of the key antibody growth areas of the last 20 years.
Quadromas
Arguably, the first bispecific antibodies – two conjugated antibodies of differing specificity – were described in 1961 (1), but it wasn’t until after the advent of monoclonal antibodies in 1975 that interest in bispecific antibodies, especially as potential therapeutics, began to take off. Milstein, in 1983, went on to describe a method for producing bispecific antibodies through the somatic fusion of two hybridoma cell lines, forming a quadroma cell – sometimes referred to as a hybrid hybridoma – capable of secreting whole IgG antibodies with the binding characteristics of the two individual parental hybridomas in a single molecule (2). This process isn’t flawless. Due to the random pairing of heavy and light chains in the quadroma a number of different miss-paired immunoglobulin species are produced, only one of which is the desired functional bispecific antibody. This is shown in the figure below. The desired functional bispecific antibody is only 12.5% of the theoretical population of antibodies making production of bispecific antibody in large quantities and to high purity very challenging. Despite this one bispecific antibody, catumaxomab (trade name Removab®) an anti-CD3 and EpCAM bispecific antibody (3), has reached clinical approval. Production was facilitatated by having one chain as a rat IgG2b and the other as mouse IgG2a causing species restricted pairing. Protein A purification with sequential pH elution enables separation of the different paired formats.
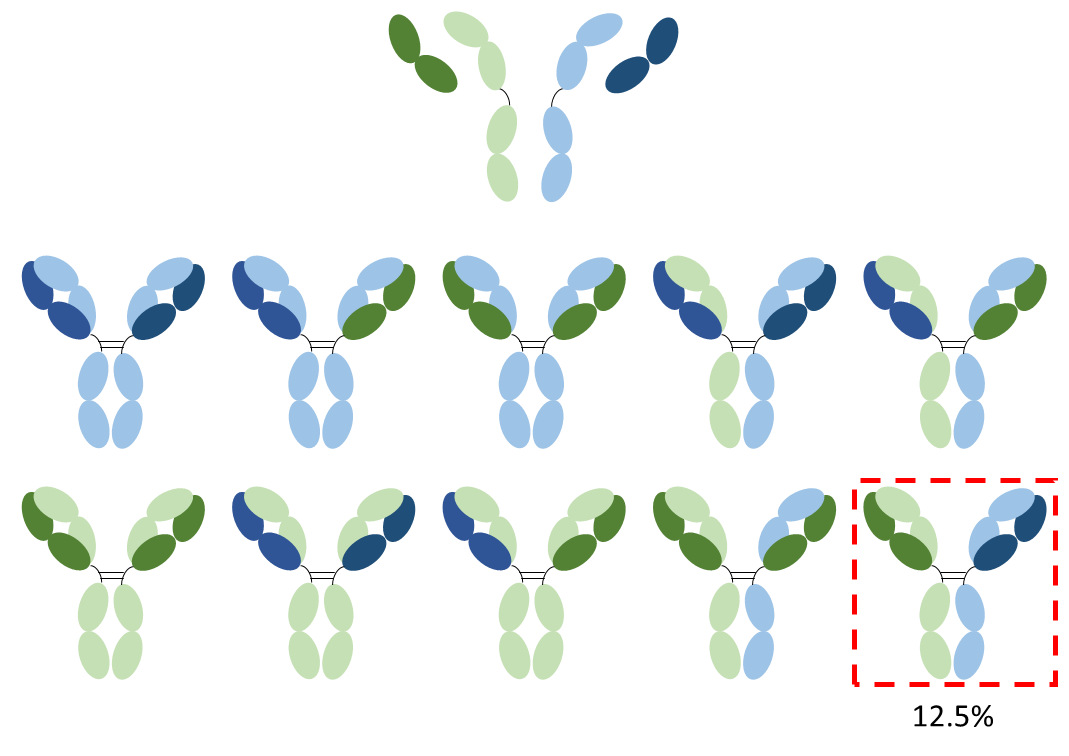
Figure 1. The chain association problem. Representation of the antibody combinations that can be produced by a quadroma cell line assuming random chain association. In total 16 formats are possible, of which six are identical. Six tetramers, including the desired bispecific antibody, occur twice (each with a yield of 12.5%) and four tetramers occur once (each with a yield of 6.25%).
In the last 20 years efforts have focused on recombinant DNA technology to enable engineering of antibody formats that minimize or completely avoid this chain association problem and the remainder of this section will focus on these.
Heterodimeric bispecific antibodies
When people refer to bispecific antibodies they are typically referring to heterodimeric antibodies, i.e. a traditional IgG molecule but with one arm targeting one antigen and the other are targeting a second antigen as shown in the figure below. These molecules are thus bispecific and monovalent.
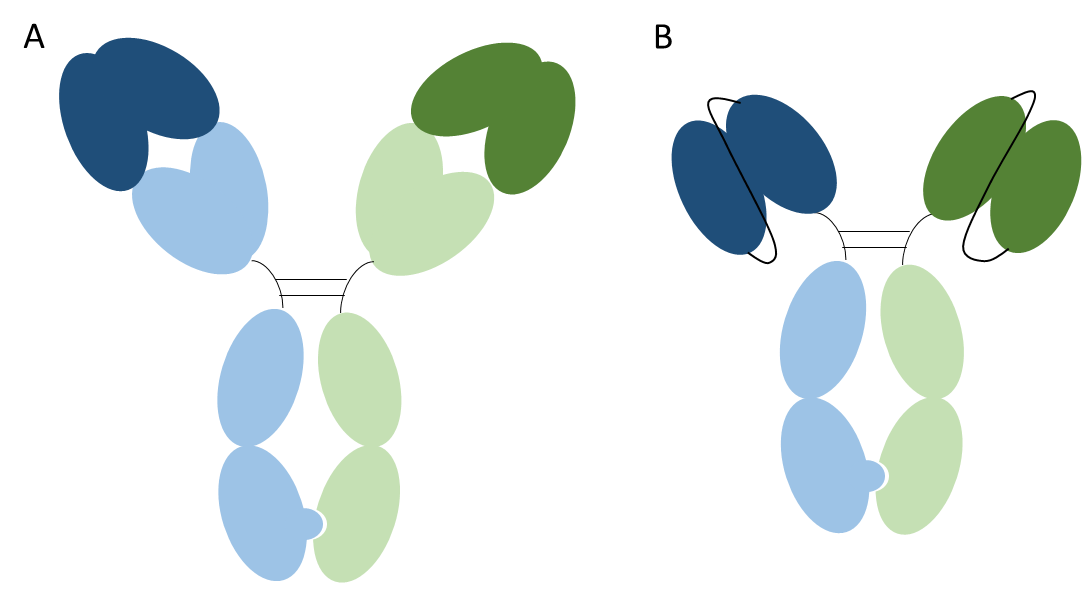
Figure 2. Heterodimeric bispecific antibodies. Image A shows a heterodimeric IgG and image B shows a heterodimeric scFv-Fc. One heavy chain and light chain pair is shown in blue and the other in green. The variable domains are shown in a darker shade. The Fc domain contains mutations that result in the preferential formation of heterodimer. Both formats shown are bispecific and monovalent.
The first bispecific antibody of this kind was described by Genentech (4). They created a “knobs-into-holes” antibody (often abbreviated as KiH) whereby a large amino acid on one chain was replaced with a small amino acid (mutation Y407T) and on the second chain a small amino acid was replaced with a large (mutation T366Y). This resulted in preferential formation of heterodimer. Further mutations were later reported by this group to enhance heterodimer formation further (5).
Subsequent attempts to generate heterodimeric bispecific antibodies have followed a similar approach. Amgen have created heterodimers by altering the charge of the Fc domain interface, or as they call it electrostatic steering (6). EMD Sereno have created SEEDbodies consisting of alternating human IgA and IgG beta strand sequences to generate asymmetric but complementary domains (7).
These approaches and many similar ones suffer from two main problems: i) light chain shuffling; and ii) homodimer formation. The aforementioned approaches address the issue of heavy chain association but it is also essential to ensure correct light chain association. If this is not addressed the light chains can pair with either heavy chain resulting in a mixture of species. This can be resolved by using antibodies that have been developed to have a common light chain (8) but in most situations this option is not available. The most successful engineering approach to resolve this problem has come from Roche with the development of CrossMab (9). In this format the VH-VL or CH1-CL domains are exchanged by domain crossover between the heavy and light chain Fab domains, resulting in preferential heterodimer formation.
Alternatively, the light chain shuffling problem can be removed completely by switching to a scFv-Fc format (Figure 2B). However, all of these approaches to force heterodimer formation still result in a varying amount of homodimer being present, which must be removed during purification. The amount of the preferred heterodimer varies from approach to approach but some mutations are claimed to result in >95% heterodimer. However, to achieve this care must be taken on ensure comparable expression levels between the two different chains.
A completely unrelated approach has been developed by Genmab and is based on the occurrence of Fab-arm exchange in IgG4s (10, 11). In this so-called Duobody approach two homodimeric antibodies are expressed individually and purified. Under redox conditions the two antibodies are then mixed together and pair into a heterodimeric antibody. As the light chains are already correctly paired this removes the light chain shuffling problem and reported levels of heterodimer formation are high.
Bispecific antibody fusions
In an effort to avoid the main manufacturing issues associated with heterodimeric bispecific antibodies (i.e. light chain shuffling and homodimer formation) groups have looked into IgG fusions. The direct genetic coupling of the desired variable domains in these formats avoids production of alternative shuffled variants but results in a non-standard IgG molecule which itself may cause manufacturing problems such as reduced expression levels or increased aggregation tendency.
Abbott have developed the dual variable domain antibodies (DVD-Ig) (12) with a very similar independent approach also taken by Sanofi to generate tetravalent bispecific tandem immunoglobulins (TBTI). In these formats an IgG is elongated at its N-terminus on the corresponding heavy and light chains by an additional variable domain of a second antibody (Figure 3A). Thus the resulting IgG molecule is a heterotetramer composed of two heavy and two light chains, with the variable domains orientated in a tandem configuration.
An alternative approach is the fusion of a scFv at the C-terminus of the antibody, thus generating a molecule that binds one antigen at the N-terminus of the molecule and a second antigen at the C-terminus (Figure 3B). In a similar manner a scFv-Fc-scFv format could also be engineered (Figure 3C). These formats are all bispecific and bivalent.
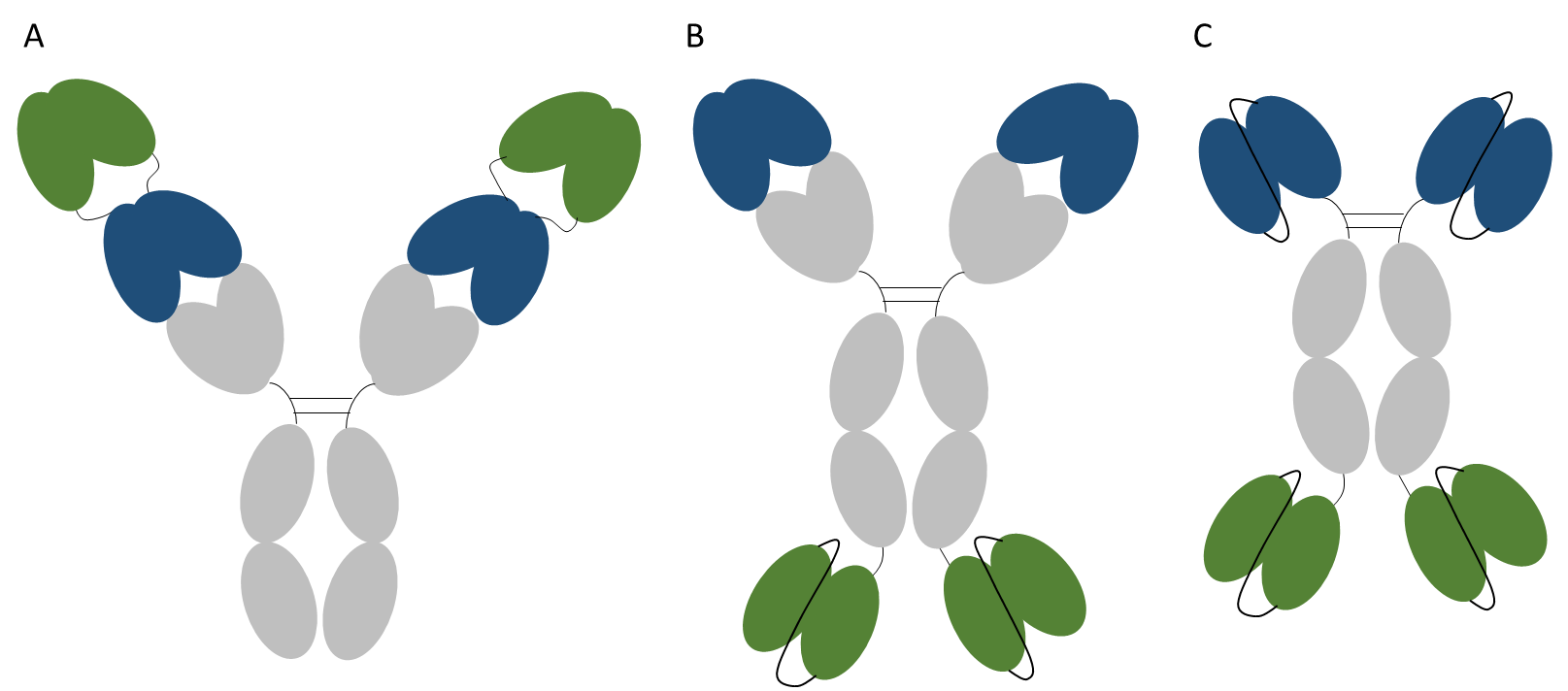
Figure 3. Bispecific antibody fusions. Representations of a dual variable domain antibody (A), an IgG-scFv (B) and a scFv-Fc-scFv (C). In each format one pair of variable domains is shown in blue and the second pair in green. All formats shown are bispecific and bivalent.
Bispecific antibody fragments
A whole zoo of multi-specific antibody fragments have been developed over the last 30 years. A small selection of these are shown on the antibody fragments section of this resource and many more are reviewed elsewhere (see 13-17).
Although numerous formats are currently in clinical development the only approved bispecific antibody fragment to date is blinatumomab (trade name Blincyto®) from Amgen. This is a bispecific T-cell engager or BiTE. BiTEs are constructed by genetic fusion of two single chain variable fragments (scFvs). Once scFv binds to CD3e on T-cells and the other binds to a surface molecule on the specific target cancer cells. Once bound to the target antigen on the cancer cell, binding of CD3 leads to activation and polyclonal expansion of cytotoxic T-cells ultimately resulting in lysis of tumour cells (18).
Multi-specific antibodies
As described in the bispecific antibody fragments section, a whole plethora of multi-specific antibody fragments exist. For the purpose of this section these will be ignored and only IgG-like multi-specific antibodies will be considered. Confusingly bispecific antibodies targeting two different antigens are sometimes referred to as trispecific if they also contain an Fc domain that interacts with Fc receptors. For the purpose of this resource we consider such antibodies to be bispecific.
As one would expect, multi-specific antibodies can be generated by any combination of the numerous technologies previously described to generate tri, tetra and penta specific antibodies with varying valencies. MedImmune have reported the use of an IgG with back-to-back scFvs at the C-terminus (19). Equally a format consisting of scFvs fused at either end of the IgG could be easily imagined.
Another approach to generating multi-specific antibodies comes from Zyngenia, with their Zybody platform. This technology incorporates antigen binding peptides at some or all of the N and C-termini of antibody heavy and light chains, resulting in up to five specificities (20).
<< Fc engineering Antibody drug conjugates >>
References
An ever advancing topic such as this is too large to cover in any single review. We highly recommend reading papers 13-17 in this list which cover many topics not on this resource page and also cover some of the areas described in this resource in more detail.
- Nisonoff, A., and Rivers, M.M. (1961). Recombination of a mixture of univalent antibody fragments of different specificity. Arch. Biochem. Biophys. 93, 460–462.
- Milstein, C., and Cuello, A.C. (1983). Hybrid hybridomas and their use in immunohistochemistry. Nature 305, 537–540.
- Chelius, D., Ruf, P., Plöscher, M., Liedtke, R., Gansberger, E., Hess, J., Wasiliu, M., and Lindhofer, H. (2010). Structural and functional characterization of the trifunctional antibody catumaxomab. MAbs 2, 309–319.
- Ridgway, J.B., Presta, L.G., and Carter, P. (1996). “Knobs-into-holes” engineering of antibody CH3 domains for heavy chain heterodimerization. Protein Engineering 9, 617–621.
- Merchant, A.M., Zhu, Z., Yuan, J.Q., Goddard, A., Adams, C.W., Presta, L.G., and Carter, P. (1998). An efficient route to human bispecific IgG. Nat Biotech 16, 677–681.
- Gunasekaran, K., Pentony, M., Shen, M., Garrett, L., Forte, C., Woodward, A., Ng, S.B., Born, T., Retter, M., Manchulenko, K., et al. (2010). Enhancing antibody Fc heterodimer formation through electrostatic steering effects: applications to bispecific molecules and monovalent IgG. J. Biol. Chem. 285, 19637–19646.
- Muda, M., Gross, A.W., Dawson, J.P., He, C., Kurosawa, E., Schweickhardt, R., Dugas, M., Soloviev, M., Bernhardt, A., Fischer, D., et al. (2011). Therapeutic assessment of SEED: a new engineered antibody platform designed to generate mono- and bispecific antibodies. Protein Eng. Des. Sel. 24, 447–454.
- Jackman, J., Chen, Y., Huang, A., Moffat, B., Scheer, J.M., Leong, S.R., Lee, W.P., Zhang, J., Sharma, N., Lu, Y., et al. (2010). Development of a two-part strategy to identify a therapeutic human bispecific antibody that inhibits IgE receptor signaling. J. Biol. Chem. 285, 20850–20859.
- Schaefer, W., Regula, J.T., Bähner, M., Schanzer, J., Croasdale, R., Dürr, H., Gassner, C., Georges, G., Kettenberger, H., Imhof-Jung, S., et al. (2011). Immunoglobulin domain crossover as a generic approach for the production of bispecific IgG antibodies. Proc. Natl. Acad. Sci. U.S.A. 108, 11187–11192.
- Labrijn, A.F., Meesters, J.I., de Goeij, B.E.C.G., van den Bremer, E.T.J., Neijssen, J., van Kampen, M.D., Strumane, K., Verploegen, S., Kundu, A., Gramer, M.J., et al. (2013). Efficient generation of stable bispecific IgG1 by controlled Fab-arm exchange. Proc. Natl. Acad. Sci. U.S.A.
- Labrijn, A.F., Meesters, J.I., Priem, P., de Jong, R.N., van den Bremer, E.T.J., van Kampen, M.D., Gerritsen, A.F., Schuurman, J., and Parren, P.W.H.I. (2014). Controlled Fab-arm exchange for the generation of stable bispecific IgG1. Nat Protoc 9, 2450–2463.
- Gu, J., and Ghayur, T. (2012). Generation of dual-variable-domain immunoglobulin molecules for dual-specific targeting. Meth. Enzymol. 502, 25–41.
- Holliger, P., and Hudson, P.J. (2005). Engineered antibody fragments and the rise of single domains. Nat. Biotechnol. 23, 1126–1136.
- Chan, A.C., and Carter, P.J. (2010). Therapeutic antibodies for autoimmunity and inflammation. Nat Rev Immunol 10, 301–316.
- Cuesta, Á.M., Sainz-Pastor, N., Bonet, J., Oliva, B., and Álvarez-Vallina, L. (2010). Multivalent antibodies: when design surpasses evolution. Trends in Biotechnology 28, 355–362.
- Byrne, H., Conroy, P.J., Whisstock, J.C., and O’Kennedy, R.J. (2013). A tale of two specificities: bispecific antibodies for therapeutic and diagnostic applications. Trends Biotechnol. 31, 621–632.
- Spiess, C., Zhai, Q., and Carter, P.J. (2015). Alternative molecular formats and therapeutic applications for bispecific antibodies. Molecular Immunology 67, 95–106.
- Stieglmaier, J., Benjamin, J., and Nagorsen, D. (2015). Utilizing the BiTE (bispecific T-cell engager) platform for immunotherapy of cancer. Expert Opinion on Biological Therapy 15, 1093–1099.
- Dimasi, N., Fleming, R., Hay, C., Woods, R., Xu, L., Wu, H., and Gao, C. (2015). Development of a Trispecific Antibody Designed to Simultaneously and Efficiently Target Three Different Antigens on Tumor Cells. Mol. Pharmaceutics 12, 3490–3501.
- LaFleur, D.W., Abramyan, D., Kanakaraj, P., Smith, R.G., Shah, R.R., Wang, G., Yao, X.-T., Kankanala, S., Boyd, E., Zaritskaya, L., et al. (2013). Monoclonal antibody therapeutics with up to five specificities: functional enhancement through fusion of target-specific peptides. MAbs 5, 208–218.

 United Kingdom (UK)
United Kingdom (UK)