Due to their high specificity and selectivity antibodies have always had the potential to be of great use as biochemical tools for a range of applications including selection, identification, purification and as therapeutics. Broadly speaking antibodies are categorized into two groups (polyclonal or monoclonal) and are utilized in three main areas (research, diagnostics and therapeutics).
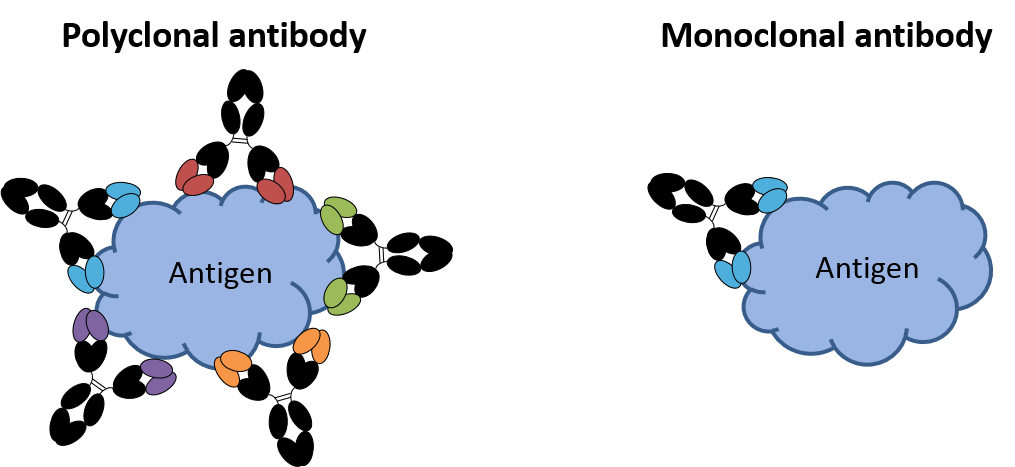
Figure. Categories of antibodies.
Comparison of polyclonal antibodies, which bind to the same antigen but different epitopes, with monoclonal antibodies which all bind to the same epitope on a target antigen.
Polyclonal antibodies (pAbs) are a heterogeneous mixture of antibodies directed against various epitopes on the same antigen. The antibodies are generated by different B-cell clones of the animal and as a consequence are immunochemically dissimilar, with different specificities and affinities.
The true potential of antibodies as specific targeting agents was not realized until ground breaking work by Köhler and Milstein in 1975 resulted in the production of monoclonal antibodies or mAbs (1). These were produced by the fusing of antibody producing mouse spleen cells with an immortal mouse myeloma cell line, resulting in the formation of an immortal cell line, known as a hybridoma, expressing a single antibody with specificity for one particular epitope on an antigen, i.e. a monoclonal antibody. Once produced hybridomas can be cultured in vitro indefinitely allowing the relatively easy purification of large quantities of monoclonal antibody.
Although a hybridoma-derived monoclonal antibody is characterized for function, the exact sequence of the antibody is unknown. A recombinant antibody is a type of monoclonal antibody where the sequence has been identified and then produced synthetically; for example, in human embryonic kidney (HEK) or Chinese Hamster Ovary (CHO) cells. As recombinant antibodies are defined at the sequence level, you can guarantee production of a truly monoclonal antibody with no batch-to-batch variation. Absolute Antibody offers recombinant antibody production as a service, as well as a catalog of engineered recombinant antibodies.
Research
Antibodies are vital tools in many of the laboratory techniques that are used to answer basic research questions. Due to their outstanding specificity they make exquisite tools that allow researchers to identify molecules that cannot be seen by the naked eye and thus enable conclusions to be drawn about the target molecule and pathway of interest. Routine procedures such as western blot, flow cytometry, immunohistochemistry (IHC), enzyme-linked immunosorbent assay (ELISA) and many others all rely antibodies.
Diagnostics
Antibodies have become a critical component of many diagnostic assays. Uses included but are not limited to the detection of infections, recognition of allergies and the measurement of hormones and other biological markers in blood.
Therapeutics
The ability of antibodies to bind an almost unlimited number of target proteins with high specificity always meant they were destined to be used as therapeutics. As early as 1900 Paul Ehrlich coined the term ‘magic bullets’ in reference to antibodies. Following the ground breaking publication on the production of monoclonal antibodies (1) the early success of the first therapeutic antibody OKT3 (muromonab) as a treatment for transplant rejection was not immediately followed by the wave of approvals that many anticipated. However, since the mid to late 1990s therapeutic antibodies have become one of the fastest growing classes of therapeutics in the biological drugs market (2). Some of the work that led to this breakthrough will be discussed in more detail in the antibody engineering section.
<< Other antibody interactions Antibody engineering >>
References
- Köhler, G., and Milstein, C. (1975). Continuous cultures of fused cells secreting antibody of predefined specificity. Nature 256, 495–497.
- Pavlou, A.K., and Belsey, M.J. (2005). The therapeutic antibodies market to 2008. Eur J Pharm Biopharm 59, 389–396.

 Germany
Germany