Alzheimer’s disease (AD) is a chronic neurodegenerative disorder affecting more than 50 million people worldwide. AD is the most common age-associated dementia in the world and is characterized by progressive cognitive impairment in memory and functional dysfunction. Because of the continuous growth of the aging population and increasing life expectancy, further research aimed at early diagnosis and modifying the course of the disease is needed.
To aid in the effort of developing diagnostic and therapeutic approaches for AD, Absolute Antibody offers an unmatched collection of recombinant anti-amyloid beta monoclonal antibodies (Table below).
Amyloid Beta and Alzheimer’s Disease
Clinical symptoms of AD primarily involve cognitive dysfunction, such as learning abilities, and memory loss. AD patients suffer from progressive disability due to dementia and movement disorders. The disease is fatal on average within four to eight years from the diagnosis (Song et al., 2022). According to the current theories, AD pathology is driven by both modifiable and inherited risk factors. The modifiable risk factors include disorders such as diabetes, hypertension and cardiovascular diseases, and unhealthy lifestyle, while aging and genetic factors contribute to inherited risk factors (Song et al., 2022).
Pathologically, AD is defined as a neurodegenerative disorder characterized by the accumulation of amyloid-beta (Aβ) plaques, abnormal aggregated forms of the Aβ-proteins, in the spaces between nerve cells, and intracellular accumulation of hyperphosphorylated tau proteins leading to the formation of neurofibrillary tangles (Shi et al., 2022). The amyloid hypothesis, proposed by Hardy and Allsop and updated by Selkoe, holds that the accumulation of Αβ into soluble oligomeric and/or insoluble amyloid deposits is the initial event which triggers the underlying pathogenesis of AD (van Dyck et al., 2017). Therefore, it is believed that the clearance of brain Aβ plaques may treat AD and cease disease progression. Because of their selectivity and specificity, anti-amyloid monoclonal antibodies are particularly promising candidates to reverse the amyloid plaque pathology (Bateman et al., 2022). However, early treatment start is deemed beneficial as impairments of the meningeal lymphatic system following disease onset, or old age, reduce treatment efficacy, as demonstrated in a Nature paper for which Absolute Antibody custom manufactured a recombinant amyloid beta antibody and accompanying isotype control (Da Mequita et al., 2021).
Up to now, the FDA has approved two amyloid beta monoclonal antibodies for treatment of Alzheimer’s disease, aducanumab and lecanemab. These therapeutic antibodies differ considerably in their relative affinities for protofibrils or fibrils, as shown in another paper for which Absolute Antibody recombinantly produced an amyloid beta-directed antibody (Söderberg et al., 2022). Among them, lecanemab, a humanized IgG1 version of the mouse mAb158, is a promising treatment for AD, due to the reduction of brain Aβ levels, a small effect in alleviating cognitive decline in early stages of the disease, and better safety. However, clinical trials of this antibody have not been without setbacks.
Recombinant Engineered Antibodies
The Absolute Antibody catalog offers three amyloid beta antibody clones: WO2, 6E10 and 21F12. As with all our catalog antibodies, the anti-Aβ clones are recombinantly produced to ensure batch-to-batch reproducibility and enable antibody engineering. The antibodies are available off-the-shelf in different engineered formats, including mouse, rabbit and human species, as well as various isotypes and fragments. The antibodies can also be custom engineered into other formats upon request.
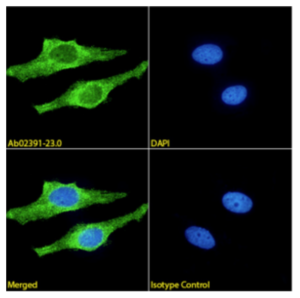
Further, Absolute Antibody confirmed the specificity of the recombinantly produced clones. Figure 1 compares the recombinant and hybridoma-derived WO2 (Ab00239) clone in Western blot analysis and in an assay detecting immobilized amyloid-beta derived peptides. Figure 2 shows immunohistochemical and Western blot analysis for 6E10 (Ab00714) and Figure 3 shows IF staining for clone 21F12 (Ab02391).
The epitopes, together with the reported applications and formats currently available on the website, are depicted in the table below. Please contact us if you are interested in the anti-amyloid beta antibodies, their applications, or their availability in different formats. With our custom services, you can tailor your approach to exactly what you need. We offer antibody engineering in every format, optimized antibody expression via our recombinant system, and sequencing to protect your work.
In addition, be sure to check out available Alzheimer’s disease research reagents from other brands within our parent company Absolute Biotech: goat polyclonal antibodies from Everest Biotech; antibodies and cell lines from Kerafast; and antibodies, proteins and ELISA kits from LSBio.

Fig. 1. (A) Coomassie blue-stained SDS-PAGE gel comparing Absolute Antibody’s recombinant WO2 (Ab00239) with antibody from the corresponding hybridoma under native and reducing (DTT) conditions. Both preparations showed, by this method, comparative purity. (B) The specificity of the antibodies was tested in assays detecting immobilized peptides. (C) Various amyloid-beta-derived peptides were probed with WO2 (50 ng/ml) and a goat-anti-mouse peroxidase-conjugated tracer. The recombinant and hybridoma-derived antibody showed comparable selectivity; however, higher signal intensity was observed for the recombinant material. Titration of recombinant and hybridoma-derived WO2 against Aß 2-42 (D) and Aß 3-42 (E) indicates that the recombinant antibody has higher activity than the hybridoma-derived counterpart.
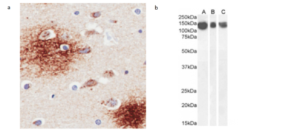
Fig. 2. (A) Immunohistochemical staining of human Alzheimer’s disease hippocampus tissue using 6E10 (Ab00714). (B) Western blot using 6E10 (Ab00714) on human brain cerebral cortex (A), frontal cortex (B) and cerebellum (C) lysate samples.
| Catalog Number | Clone Name | Antigen | Applications | Formats |
|---|---|---|---|---|
| Ab00239 | WO2 | Amyloid beta (aa 4-10) | IHC-P, WB, ELISA | Rabbit IgG Mouse IgG2a |
| Ab00714 | 6E10 | Anti-Amyloid Beta | IP, WB, ELISA, EM, IHC | Rabbit IgG Mouse IgG1 Mouse Fab fragment Mouse F(ab)2 Mouse IgG2a |
| Ab02391 | 21F12 | Anti-Amyloid Beta | IP, WB, ELISA, IHC | Rabbit IgG Human IgG1 Mouse IgG2b |
Article compiled by Dr. Lucia Lupica-Spagnolo, Absolute Antibody Product Development Manager
Latest News
Upcoming Events
Please join us at the following conferences and events. Stop by our booth, or get in touch to arrange a meeting.
See All Dates
 United Kingdom (UK)
United Kingdom (UK)