In vivo studies in mouse models are crucial to holistic understanding of immune function. With the ever-rising interest in antibodies as therapeutics – the FDA has approved more than 100 monoclonal antibody products, with many more in review – antibodies and other tools for in vivo mouse model research have never been more important.
A steppingstone on the clinical pathway, in vivo studies in mouse models have plentiful benefits. Researchers can track systemwide immunological responses to treatments in a mammalian model system similar to humans, offering important preclinical insight into the efficacy of potential drug candidates.
But there are confounding factors to in vivo studies that can stop experiments in their tracks. When using antibodies in murine models, some are more immunogenic than others. Highly unrelated antibody species, high endotoxin contaminant levels, and reagent preservatives can elicit immune responses in mouse models that reduce the efficacy of antibodies and create nonspecific background noise, resulting in muddy data.
Antibody Solutions for Improved In Vivo Research
Our VivopureX™ line of recombinant antibodies has been designed specifically for improved in vivo research in mouse models. Many of the most popular antibody clones are sourced from rats or hamsters, which trigger unwanted immune responses in mice. In contrast, our VivopureX™ antibodies are chimerized to have a mouse Fc region while keeping the original variable antigen-binding region intact. This reduces immunogenicity in mouse models, meaning antibodies work for longer and cohorts respond more consistently. VivopureX™ antibodies also have high purity (>98%) and industry-leading low endotoxin levels (<0.5 EU/mg, versus the <1 EU/mg or even <2 EU/mg standard for many reagent antibodies).
Data show the benefits of our antibodies’ low immunogenicity and high purity: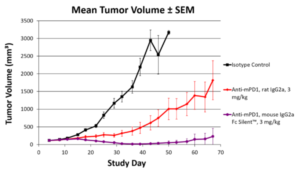
- University of Basel research demonstrates that the species-matched mouse-anti-mouse VivopureX™ version of an antibody can deplete CD8+ T-cells more completely and for longer in mice than the original rat form of the antibody.
- Our research poster illustrates that syngeneic mouse IgG2a Fc Silent™ PD-1 antibodies reduce tumor size in mouse models more effectively than traditional rat monoclonal antibodies.
- A recent paper from the University Medical Center Utrecht found that our VivopureX™ mouse-anti-mouse Ly-6G antibody was able to deplete neutrophils in mice more efficiently than the original rat Ly-6G antibody.
Clinically Relevant Engineered Antibody Formats
A signature aspect of our VivopureX™ antibodies is their recombinant production. In addition to providing the utmost in batch-to-batch reproducibility, recombinant production means we have full control over the Fc region and therefore effector function. We have engineered each VivopureX™ antibody’s effector function to fit the needs of today’s researchers, offering formats that are the most clinically relevant.
PD-1: The target of seven approved or under-review antibody therapeutics, programmed cell death protein 1 (PD-1) is a huge topic of interest in the immunotherapy community. The aim of anti-PD-1 antibodies is to block the activity of PD-1 without killing the T cells important to immune function. We therefore offer our VivopureX™ anti-PD-1 antibodies with Fc Silent™ engineering. Our Fc Silent™ mutation abrogates binding of Fc receptors and removes effector function in vivo, making our PD-1 antibodies ideal for blocking applications.
Our recombinant Fc Silent™ VivopureX™ PD-1 antibody has been used in conjunction with a monoclonal antibody targeting the GARP:TGF-β1 protein complex to overcome anti-PD-1 resistance and promote anti-tumor activity in mice. The data show that the anti-GARP:TGF-β1 antibody administered with Absolute Antibody’s PD-1 antibody resulted in a higher proportion of mice completely rejecting their tumors (de Streel, 2020; Bertrand, 2021). For research on the anti-tumor properties of PD-1 antibodies, our line houses clinically relevant recombinantly engineered formats.
CTLA-4: Our VivopureX™ line includes antibodies that block cytotoxic T-lymphocyte associated protein 4 (CTLA-4). CTLA-4 is the target of FDA-approved cancer-directed monoclonal antibody therapeutics, namely ipilimumab and tremelimumab. Our anti-CTLA-4 antibodies are available in the VivopureX™ collection as mouse IgG2a subtypes, with and without our proprietary Fc silencing engineering. Our choice to provide the mIgG2a subtype of this antibody stems from evidence that suggest IgG2a anti-CTLA-4 antibodies have stronger anti-tumor activity via depletion of regulatory T cells (Tregs) in tumors than the IgG2b subtype of the same antibody (Selby et al., 2013). Additionally, evidence from Selby et al. shows that Fc-silenced isotypes lead to peripheral Treg expansion compared to their Fc-functioning counterparts. Further research, like this recent study, suggest there are Fc-independent anti-tumor functions in anti-CTLA-4 antibodies. To realize the full breadth of potential CTLA-4-directed antibody therapies, we provide this antibody in Fc Silent™ and functioning Fc region formats in the most clinically-promising subtype.
TIGIT: Immune checkpoint inhibitors on the rise, anti-TIGIT (T cell immunoreceptor with Ig and ITIM domains) antibodies are promising tools to study new cancer therapeutic options. With clones that can induce antagonistic (anti-TIGIT [1B4]) and agonistic effects (anti-TIGIT [4D4]) on the immune system’s reaction to antigens, TIGIT is a multi-faceted antibody target that can be used to study cancer therapeutics and autoimmune diseases alike. An in vivo mouse and human study from our customers used VivopureX™ anti-TIGIT antibodies to investigate the role of TIGIT and its ligand CD155 in inflamed tumor microenvironments. The data suggest that the TIGIT/CD155 axis affects resistance to other immune checkpoint inhibitors. As more strategies targeting inhibitory immune checkpoints are brought to the clinic (some with disheartening results, like Merck’s TIGIT-based anti-cancer therapy that recently failed its phase 2 trial), further research and development of TIGIT as an anti-cancer therapy is needed.
Additionally, new data illustrates that there are isotype-dependent factors that affect antibody anti-tumor activity (Preillon et al., 2021). In this study, murine anti-TIGIT IgG2a antibodies isotypes (known to effectively engage Fcγ receptors) were found to be more effective at inhibiting tumor growth and reducing tumor size than their IgG1 isotype (which show reduced affinity for FcγR activation) counterparts. Another study further elucidates the unique function of the IgG2a anti-TIGIT isotype, revealing that it may affect anti-tumor activity by engaging FcγRs on myeloid cells stronger than IgG1 antibodies, inducing reverse activation signals (Han et al., 2020). These new views of the mechanisms of action and of the role anti-TIGIT isotypes play in therapeutic applications means more tools are needed. To address this need, our antibody experts engineered the VivopureX™ anti-TIGIT antibodies into the mouse IgG2a isotype, which joins our original mouse IgG1 isotype in the collection.
Our high purity, 100% defined VivopureX™ recombinant antibodies are designed to help researchers bridge the gap in understanding TIGIT with the best mouse in vivo tools, available in a variety of isotypes to span the full range of anti-TIGIT possibility.
We’re Here to Help
Explore our VivopureX™ catalog and see how our antibodies can open up doors for your murine in vivo research. Absolute Antibody specializes in animal-free recombinant production of antibodies, and our engineers can turn any antibody—whether from our catalog or your lab’s own antibody—into the format you need. Contact us and get connected with antibody engineers and experts to solve your research problems, so you can get back to moving your projects along and accelerating research in your field.
Latest News
Upcoming Events
Please join us at the following conferences and events. Stop by our booth, or get in touch to arrange a meeting.
See All Dates
 United Kingdom (UK)
United Kingdom (UK) 