Epitope tagging is a method of expressing proteins where an epitope for a specific antibody is fused to a target protein using recombinant DNA techniques (Brizzard et al., 2001). Epitope tagging has many applications like monitoring protein expression, localizing proteins at the cellular and subcellular levels, protein purification, as well as the analysis of protein topology, dynamics and interactions (Jarvik et al., 1998).
It is possible to detect proteins for which no antibody is available by choosing an epitope for which an antibody is available. This is especially useful for the characterization of newly discovered proteins and proteins of low immunogenicity. If you choose the appropriate epitope and antibody pair, it is possible to find a combination with properties that are suitable for the desired experimental application, such as western blot analysis, immunoprecipitation, immunochemistry, and affinity purification (Brizzard, 2008). We are able to enable increasingly multiplexed experiments by using multiple epitope and antibody pairs, especially in combination with our recombinant antibody technology.
In research, there is always a need for a positive control protein to make sure that the epitope tag-antibody pair works as expected. Absolute Antibody’s Multi-Tag control protein (Pr00106) is a genetically engineered synthetic fusion protein composed of 17 commonly used epitope tags fused together using flexible linkers. These Multi-Tag synthetic proteins are specifically engineered so that they can be used as a positive control in western/dot blots and ELISAs (Figure 1). View our additional data on our Multi-Tag control protein, including supplemental ELISA graphs and western blots, here.
You can find our full range of recombinant and chimeric anti-epitope tag monoclonal antibodies on our website here. If you have any questions, don’t hesitate to contact us.
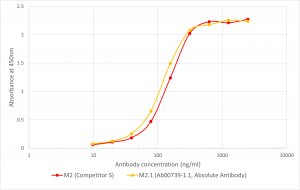
Figure 1. Binding of Anti-DDDDK antibody to Multi-Tag protein. Comparison of the binding of the anti-DDDDK antibody M2.1(Ab00739-1.1) and an equivalent antibody M2 from a competitor to Multi-Tag protein (Pr00106). Wells were pre-coated with 5 ug/mg Multi-Tag protein and then probed with serial dilutions of antibody followed by an HRP-conjugated goat anti-mouse antibody.
Latest News
Upcoming Events
Please join us at the following conferences and events. Stop by our booth, or get in touch to arrange a meeting.
See All Dates
 United Kingdom (UK)
United Kingdom (UK)