- {{heading}}
- Ab00699-1.26 Anti-H3K4me3 [304M3-B]
- Human
- Mouse scFv-Fc
- Purified
- In Stock
Recombinant monoclonal antibody to H3K4me3. Manufactured using AbAb’s Recombinant Platform with variable regions (i.e. specificity) from the phage display antibody 304M3-B.
UniProt Accession Number of Target Protein: B2R4P9
Alternative Name(s) of Target: Trimethylated Histone H3; Trimethylated Lys; Trimethylated Lys4
Immunogen: The biotinylated peptide from histone H3 ARTKQTARKSTG-GYCD. The last four amino acids GYCD were added for biotinylation purposes.
Specificity: This antibody is highly specific for Histone H3 trimethylated at lysine 4. It has been tested on human histone H3 (K562 cells), but owing to the high conservation of histones, is expected to cross-react with many other species.
Application Notes: This antibody has been used in IP-studies and works in immunofluorescence and Western Blot. In an long-neck scFv-Fc-fusion format been shown to exhibit a high degree of specificity owing to an antigen-clasping binding mechanism (see Hattori et al. 2016).
Antibody first published in:
Hattori et al. Antigen clasping by two antigen-binding sites of an exceptionally specific antibody for histone methylation. Proc Natl Acad Sci U S A. 2016 Feb 23;113(8):2092-7 PMID:26862167
Note on publication:
Describes the characterization of the specificty of this antibody through antigen-clasping.
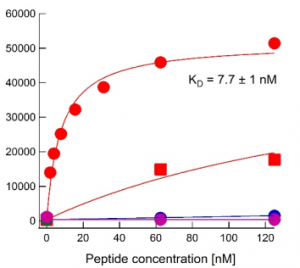
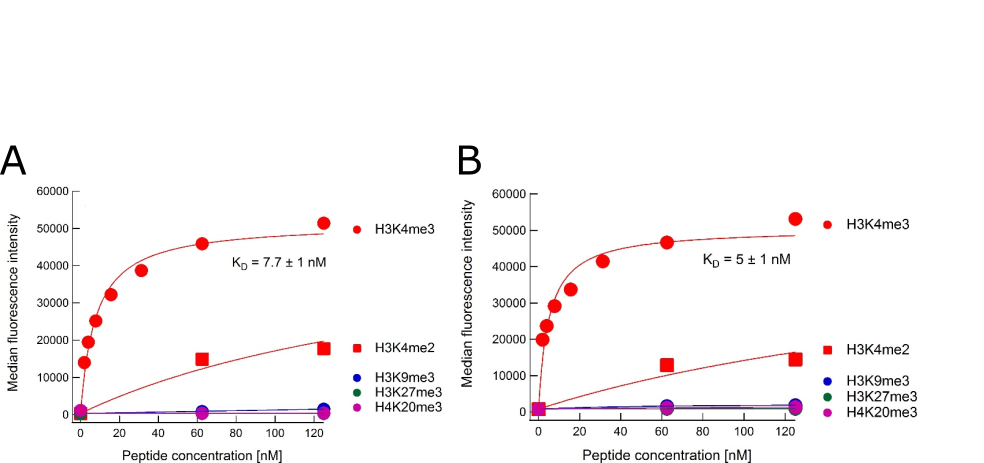
Binding curves for anti-H3K4me4 antibody Ab00699 Binding titration curves for cognate and off-target marks of the 304M3-B long-neck scFv-Fc antibody from Absolute Antibody (A, Ab00699-1.26, Lot: T1617B02) and a sample from the Koide laboratory that originally developed the antibody (B). The assay follows the peptide IP assay as described by Nishikori S, et al. (2012, PMID:23041298) and Hattori T, et al. (2016, PMID:26862167). In brief, the antibodies were loaded onto Protein G beads (for Ab00699-1.26, featuring the mouse IgG1 Fc domain) or Streptavidin beads (for the Koide lab sample featuring the mouse IgG1 Fc domain with an additional biotin moiety). With these beads immunoprecipitations were performed using different biotin-labelled peptides corresponding to modified histone peptides. Bound peptide was detected with fluorescently labelled streptavidin and quantified by flow-cytometry. Data courtesy of Dr. Takamitsu Hattori from Dr. Shohei Koide’s laboratory, formerly University of Chicago, now Perlmutter Cancer Center & Department of Biochemistry and Molecular Pharmacology, NYU Langone Medical Center.