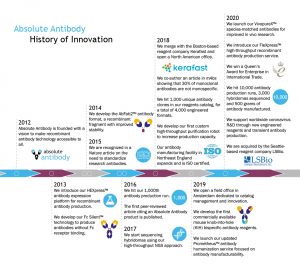
Absolute Antibody was founded in 2012 with a vision to make engineered recombinant antibodies accessible to all, and we remain the only company 100% focused on recombinant antibody technology. Though recombinant antibodies are standard in the pharmaceutical industry, these antibodies have not been as widely available for diagnostics or research. This is starting to change, however, due to the many advantages recombinant antibodies offer over traditional hybridoma-produced monoclonal antibodies.
Absolute Antibody has been at the forefront of the movement to make recombinant antibodies more readily available to all. In 2015, we were recognized in a Nature paper on the need to standardize research antibodies, and in 2018, we co-authored a mAbs Journal paper further illustrating the importance of using recombinant antibodies. Over the years, we have grown rapidly as demand for recombinant antibodies increases.
We specialize in antibody sequencing, engineering and recombinant expression, offering royalty-free custom services and a unique reagents catalog of engineered recombinant antibodies and Fc Fusion proteins. Our customers include leading pharmaceutical, biotechnology and diagnostics companies, as well as academic researchers worldwide; we have customers in 65 different countries and have sold to 14 of the top 15 pharmaceutical companies by revenue. This international customer base is served by our ISO 9001:2015-certified manufacturing facility in Northeast England and additional offices in Boston in the United States and Amsterdam in the Netherlands.
The Absolute Antibody team has deep experience in the therapeutic antibody and recombinant technology fields. Together, we have the knowledge and expertise to deliver high-quality products and services, as well as offer high-level consultation on our customers’ antibody development projects. Since our founding, we have produced more than 180 different antibody formats for our customers, as well as developed a reagents catalog of more than 15,000 engineered recombinant antibodies. For an example, check out one catalog clone (anti-EGFR, or Cetuximab) available in more than 30 different off-the-shelf formats.
In 2020, we received a Queen’s Award for Enterprise in International Trade for our rapid international expansion. To date, we have sequenced more than 5,000 hybridomas, completed more than 145 successful antibody humanization projects, and manufactured more than 23,000 different antibodies, making 1,000 grams of protein in total. We look forward to continuing this growth and working toward our vision of Engineered Recombinant Antibodies for All.
Learn more about our company history and recombinant antibody technology with our History of Innovation timeline below. View the full-size timeline here.