Antibodies of IgG isotype are the most commonly used antibodies in research, diagnostics and therapy. Upon hearing the word antibody, most of us will instantly think of those Y-shaped depictions of antibody-proteins meant to reflect the shape of IgG.
Yet nature offers a range of other formats with unique characteristics, which our recombinant antibody technology allows researchers to explore. Switching an antibody’s isotype or subtype might enable you to tailor effector function, reduce the number of needed controls, or open up new areas of investigation in allergy or autoimmunity research.
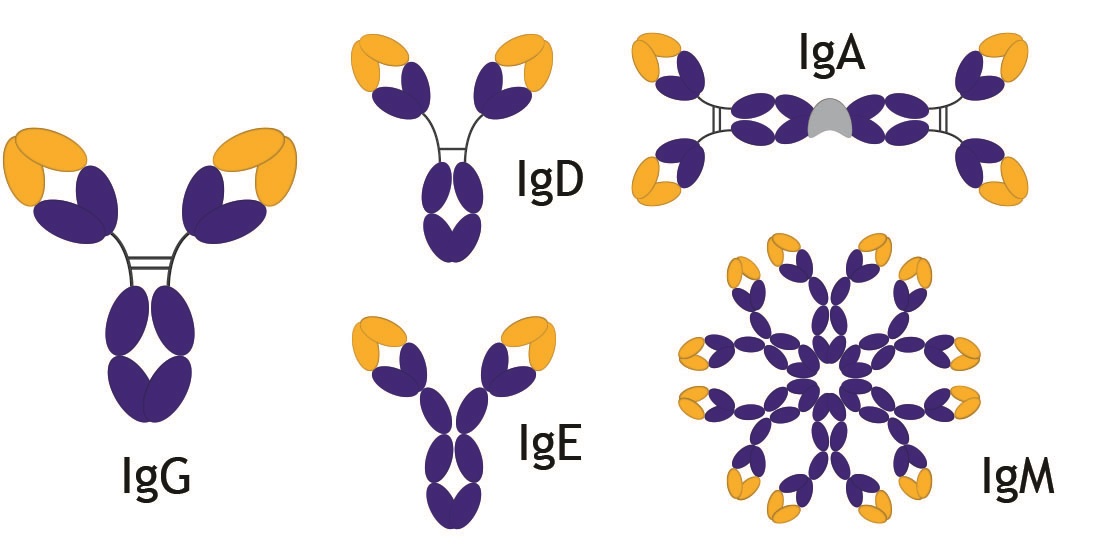 In addition to all IgG subtypes, we offer antibodies in the following isotypes:
In addition to all IgG subtypes, we offer antibodies in the following isotypes:
- IgM: the predominant antibody in the primary immune response, which can present as a pentamer or hexamer in solution and as a monomer on the surface of B-cells.
- IgA: the predominant antibody in mucous secretions such as saliva, tears, milk and intestinal juice. It is involved in preventing opportunistic pathogens from entering through mucosal barriers. It can be found in a monomeric or dimeric form.
- IgE: has two additional constant domains in place of the hinge region. It plays an important role in defense against parasites, and is also a key mediator of Type I hypersensitivity reactions or allergies.
- IgD: thought to be involved in transduction of activating signals, though other functions of IgD may still be discovered. Found predominantly on the surface of B-cells, it has a similar structure to IgG, but with an extended hinge region that is very susceptible to proteolytic digestion.
- IgY: the major immunoglobulin class in birds, reptiles, amphibians and lungfish.
Learn more about antibody isotypes and subtypes in our Antibody Resource, a free online reference resource for researchers.
Interested in a non-IgG antibody? In our recombinant antibody catalog, you can find clones engineered into different isotypes and subtypes, available for purchase off-the-shelf. We can also convert any antibody into any isotype or subtype upon request – contact us to get started!

 United Kingdom (UK)
United Kingdom (UK)