Nipah virus (NiV) has been recognized by the World Health Organization as a global health problem and included in the list of the blueprint priority diseases to research (WHO). Features indicating the pandemic potential of Nipah virus are the zoonotic nature, the ability to infect a broad range of hosts, the human-to-human transmission and the high mortality. Further, the lack of a vaccine makes it a real threat (Skowron et al., 2022).
To aid research into the virus, Absolute Antibody offers a collection of anti-Nipah virus antibodies. All antibodies are recombinantly produced to ensure batch-to-batch reproducibility, and each clone is available in a variety of engineered formats to open up new experimental possibilities. Antibodies available off-the-shelf in our reagents catalog include human IgG and IgM antibodies for use in neutralization assays and as serological controls; rabbit and mouse antibodies for co-labeling studies; and unique formats such as His-tagged Fab fragments and Fc Silent™ antibodies.
An Introduction to the Nipah Virus
The virus is named after the Malaysian village where the first outbreak was reported in 1998-1999. Since then, the virus has been responsible for outbreaks in other parts of the world, such as Bangladesh and India (Banerjee et al., 2019). The manifestation of the Nipah virus in humans is generally associated with the presence of fruit bats or flying foxes (genus Pteropus). Consumption of contaminated food, contact with infected animals and person-to-person transmission are identified as NiV transmission routes. The pathology of the disease includes encephalitis and respiratory distress, while the treatment is mostly limited to supportive care (Aditi et al., 2019; Singh et al., 2019).
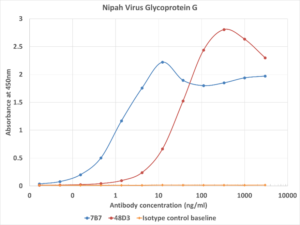
Fig. 1. ELISA activity data of antibodies 7B7 (Ab02864, blue line), 48D3 (Ab02865, red line) and an isotype control (Ab00102, orange line) binding to Nipah virus glycoprotein G recombinant protein.
NiV is grouped in the Paramyxoviridae family and the Henipavirus genus, with related viruses including Hendra virus, Cedar virus, Moijang virus and Ghana virus. It is also related to the newly identified Langya henipavirus (LayV), which was first described in the New England Journal of Medicine in August 2022 and has caused respiratory infections in China (Nature).
The Nipah virus is an enveloped paramyxovirus with negative-stranded polarity and a non-segmented RNA genome consisting of helical nucleocapsids. To its close relative within the Henipavirus genus Hendra virus (HeV), Nipah virus shows 80% similarity in terms of nucleotide homology (Thakur et al., 2019).
Nipah virus contains six genes encoding for the main structural proteins: nucleocapsid protein (N), phosphoprotein (P), matrix protein (M), fusion protein (F), glycoprotein (G) and large protein (L) or RNA polymerase. N, P and L proteins are attached to the viral RNA and form the virus ribonucleoprotein (vRNP), while F and G proteins are responsible for cellular attachment of the virion and subsequent host cell entry. For this reason, F and G proteins are the main targets of virus-neutralizing antibodies (Dang et al., 2021).
Recombinant Anti-Nipah Virus Antibodies
Absolute Antibody offers a selection of anti-Nipah virus antibodies specific for F and G proteins, as shown in the table below. The table also reports the cross-reactivity with the Hendra virus and applications of the antibodies as found in the literature. Example ELISA data for two clones 7B7 (Ab02864) and 48D3 (Ab02865) against Nipah virus glycoprotein G recombinant protein are shown in Figure 1.
The Nipah virus antibodies join our full collection of anti-virus antibodies, which also includes antibodies to detect coronavirus and other zoonotic diseases such as monkeypox, Zika, Ebola and Lyme disease. Contact us if you have any questions, if you would like to request a different isotype, species, or format, or to receive a quote.
Article complied by Dr. Lucia Lupica-Spagnolo, Absolute Antibody Junior Product Developer
| Catalog Number | Clone Name | Antigen | Cross-reactivity | Applications |
|---|---|---|---|---|
| Ab02792 | 12B2 | Nipah Virus F | Hendra virus F | Functional assay, neutralizing |
| Ab02853 | 1E11 | Nipah Virus F Site IV | Hendra virus F | IP |
| Ab02854 | 4C9 | Nipah virus F Site II | Hendra virus F | IP |
| Ab02855 | 5G7 | Nipah virus F F1 | Hendra virus F | IP, WB |
| Ab02856 | 6D3 | Nipah virus F F2 | Hendra virus F | IP, WB |
| Ab02857 | 13G5 | Nipah virus F1 | Hendra virus F | IP, WB |
| Ab02858 | 16C2 | Nipah virus F Site I | IP | |
| Ab02859 | 11F10 | Nipah virus F F2 | Hendra virus F | IP, WB |
| Ab02863 | 5B1 | Nipah virus G | Hendra virus G | IP, neutralizing |
| Ab02864 | 7B7 | Nipah virus G | Hendra virus G | IP, neutralizing, ELISA |
| Ab02865 | 48D3 | Nipah virus G | Hendra virus G | IP, WB, ELISA |
| Ab02875 | nAH1.3 | Nipah virus G | Hendra virus G | neutralizing |
Latest News
Upcoming Events
Please join us at the following conferences and events. Stop by our booth, or get in touch to arrange a meeting.
See All Dates
 Singapore
Singapore