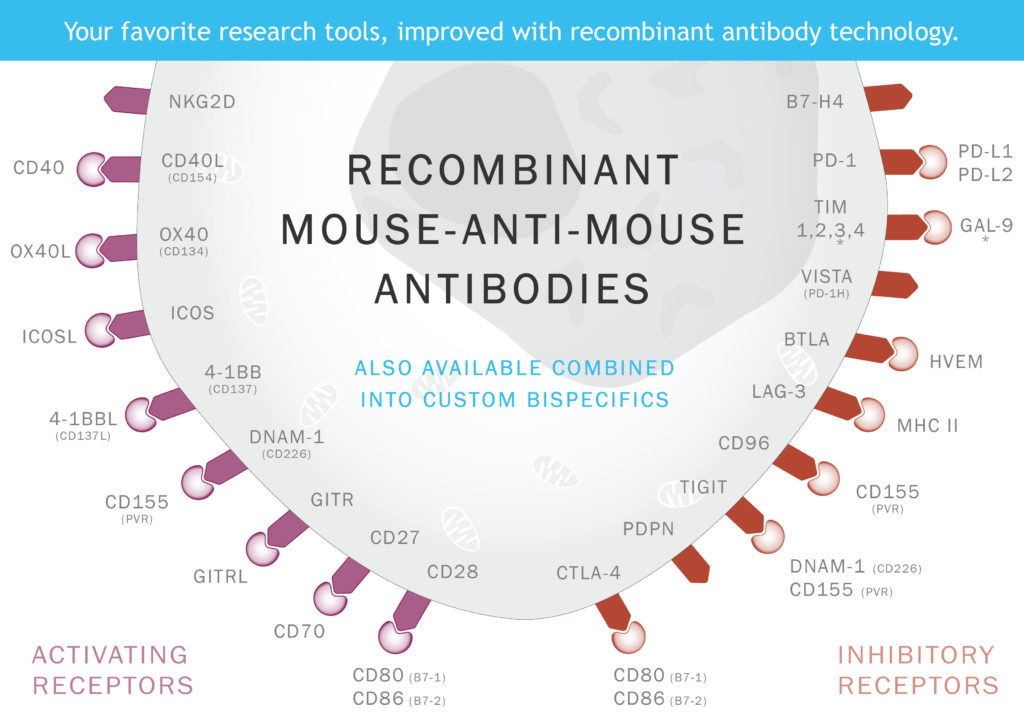
Our antibodies target many of the most clinically relevant proteins in cancer immunotherapy, including PD-1, VISTA (PD-1H), OX40, and more.
In the dynamic realm of cancer immunotherapy research, antibodies play a pivotal role in unraveling the intricacies of immune cell regulation. This article delves into the significance of cancer immunotherapy antibodies, particularly in the context of antibody therapeutics, and explores their applications in the evolving landscape of cancer research and treatment.
As the field continues to advance, with an increasing number of monoclonal antibody therapies entering clinical trials each year, researchers demand ever more advanced tools to tap into the full potential of immunotherapy. Recognizing this need, our team at Absolute Antibody has harnessed recombinant antibody technology to develop a comprehensive collection of engineered recombinant antibodies. These antibodies specifically target clinically relevant proteins, including mouse homologs of current therapeutic targets that have been engineered for optimal in vivo performance, as well as bispecific antibodies, providing researchers with cutting-edge tools to propel cancer immunotherapy research forward.
Antibodies Against Proteins in Immune Cell Regulation
The targeted development of antibodies against key proteins in immune cell regulation, including CTLA-4, PD-1, PD-L1, OX40, and GITR, holds immense promise for advancing cancer immunotherapy. These proteins, among the most searched and cited targets, play pivotal roles in modulating immune responses. Precise antibodies designed to interact with them offer groundbreaking therapeutic potential by regulating immune cell activation and function.
Understanding Cancer Immunotherapy
What is Cancer Immunotherapy?
At the forefront of advanced cancer treatments are FDA-approved monoclonal antibodies for cancer treatment, revolutionizing the field of cancer immunotherapy. Simply put, cancer immunotherapy harnesses the body’s immune system to fight cancer, and monoclonal antibody-based therapeutics play a key role in this approach. These targeted therapies are designed to specifically recognize and attack cancer cells, offering a precise and effective means of treating various forms of cancer.
How Does Cancer Immunotherapy Work?
Cancer immunotherapy operates by leveraging the body’s immune system to target and eliminate cancer cells, with monoclonal antibody therapies playing a crucial role in this process. These antibodies are designed to recognize specific proteins on cancer cells, acting as immune system boosters. For instance, FDA-approved monoclonal antibodies like pembrolizumab (Keytruda) and rituximab (Rituxan) work by blocking immune checkpoints or directly targeting cancer cells, promoting immune response and inhibiting cancer growth. This targeted approach enhances the body’s ability to identify and destroy cancer cells, marking a paradigm shift in cancer treatment strategies.
Why Immunotherapy Instead of Chemotherapy?
Immunotherapy offers distinct advantages over traditional chemotherapy, providing a targeted and precise approach to cancer treatment. Unlike chemotherapy, which can affect both cancerous and healthy cells, immunotherapy specifically harnesses the immune system to target cancer cells, minimizing collateral damage. This targeted nature of immunotherapy not only enhances treatment effectiveness but also reduces the severe side effects often associated with chemotherapy, representing a significant advancement in cancer care.
Types of Cancer Immunotherapy
Advancements in cancer care are driven by the targeted application of monoclonal antibodies in immunotherapy, showcasing tailored precision in prostate, colorectal, breast, and lung cancer treatments. Key players targeting specific aspects of cancer include antibodies against human epidermal growth factor receptor-2 (HER2), epidermal growth factor receptor (EGFR), vascular endothelial growth factor (VEGF), programmed cell death protein 1 (PD-1), and programmed cell death ligand 1 (PD-L1), underscoring the potential to revolutionize treatment strategies across these diverse cancer types.
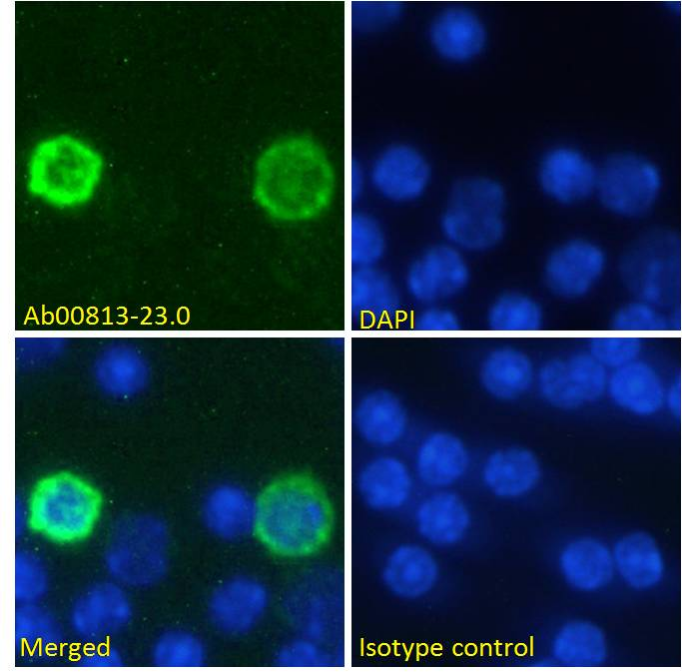
Anti-PD-1 (RMP1-14)
Immunofluorescent staining of mouse splenocytes. This antibody has been chimerized from its original rat IgG2a format to mouse IgG2a and includes Fc Silent™ engineering for tailored performance in vivo.
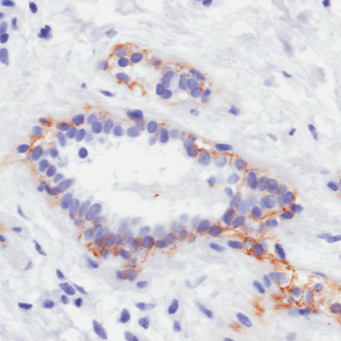
Anti-erbB-2 (Her-2/neu) [4D5-8 (trastuzumab)]
IHC staining of paraffin-embedded human breast tissue. This antibody is a recombinant research-grade biosimilar available in variety of species and formats, including the original human IgG1 and species-swapped mouse IgG2a.
Anti-TNF alpha [cA2 (Infliximab)]
Western Blot using anti-TNF alpha Human IgG1 antibody in human thyroid tumor (lane 5). This antibody is available in a variety of species and formats.
Prostate Cancer
The application of monoclonal antibodies for prostate cancer treatment showcases promising developments in immunotherapy. Notably, anti-human epidermal growth factor receptor-2 (HER2) MAb trastuzumab, anti-epidermal growth factor receptor (EGFR) MAbs cetuximab and panitumumab, and antivascular endothelial growth factor (VEGF) MAb bevacizumab have emerged as key players in targeting specific aspects of prostate cancer. Trastuzumab targets HER2-positive prostate cancer cells, while cetuximab and panitumumab focus on inhibiting EGFR, disrupting cancer cell signaling. Additionally, bevacizumab, an anti-VEGF monoclonal antibody, curtails blood supply to the tumor. These tailored applications underscore the potential of monoclonal antibodies in providing precise and effective treatments for prostate cancer.
Colorectal Cancer
The application of monoclonal antibodies for colon cancer treatment has shown promising results in the realm of immunotherapy. Specifically designed monoclonal antibodies, such as cetuximab and bevacizumab, target key proteins involved in colon cancer progression. Cetuximab, an FDA-approved monoclonal antibody, inhibits the epidermal growth factor receptor (EGFR), impeding cancer cell growth. Additionally, bevacizumab, another monoclonal antibody approved for colon cancer, targets vascular endothelial growth factor (VEGF), curtailing blood supply to the tumor. These targeted applications showcase the potential of monoclonal antibodies in providing tailored and effective treatments for colon cancer.
Breast Cancer
The use of breast cancer monoclonal antibodies in immunotherapy represents a significant advancement in breast cancer treatment. Notably, anti-human epidermal growth factor receptor-2 (HER2) MAb trastuzumab and anti-programmed cell death protein 1 (PD-1) MAb pembrolizumab have emerged as key players in targeting specific aspects of breast cancer. Trastuzumab addresses HER2-positive breast cancer, while pembrolizumab, an immune checkpoint inhibitor, activates the immune response against cancer cells. These focused applications underscore the precision and effectiveness of breast cancer monoclonal antibodies in advancing immunotherapeutic approaches.
Lung Cancer
The specific applications of immunotherapy antibodies in lung cancer treatment have brought notable advancements to the forefront of cancer care. Monoclonal antibodies such as anti-programmed cell death protein 1 (PD-1) MAb pembrolizumab and anti-programmed cell death ligand 1 (PD-L1) MAb atezolizumab have shown promise in targeting lung cancer. Pembrolizumab, as an immune checkpoint inhibitor, unleashes the body’s immune response against cancer cells by blocking PD-1, while atezolizumab targets PD-L1 to disrupt immune evasion. These precise applications of immunotherapy antibodies in lung cancer underscore their potential to revolutionize treatment strategies and improve outcomes for patients.
Empowering Cancer Immunotherapy Research
Absolute Antibody is devoted to creating the best tools for research, especially in the field of immunotherapy. Here are the product collections, educational resources, and case studies we have to power cancer immunotherapy research.
Immunotherapy Antibodies by Absolute Antibody
Absolute Antibody has spearheaded the development of a collection of recombinant engineered antibodies against clinically relevant proteins. Tools available to empower cancer immunotherapy research include:
- VivopureX™ Antibodies: These recombinant antibodies are engineered for improved in vivo performance in live mouse models, with Fc silencing and isotype switching for tailored therapeutic effects, and matched antibody species for reduced immunogenicity and longer serum half-life.
- Research-Grade Biosimilars: We offer research-grade biosimilars to grant access to research tools for the study and evaluation of biological processes without the need to source and purchase costly therapeutic-grade biologics. Our immunotherapy antibodies are engineered from the original human species into mouse, rabbit, rhesus monkey, and cynomolgus monkey formats for added research potential.
- Bispecific Antibodies: Engage two distinct targets with one antibody reagent with our range of recombinant engineered murine bispecific antibodies. Our collection offers high purity, low immunogenicity, and tailored effector function for in vivo applications in mouse models. Our antibody engineers can also custom engineer any bispecific antibody you can imagine.
- Tumor Marker Antigens: Antibodies that target tumor markers can enhance cancer treatment and diagnosis. Benefit from our selection of recombinant engineered tumor marker antibodies, with 100% biological definition, Fc Silent™ formats for eliminated ADCC, and a variety of species and isotypes available to better address your research needs.
Immunotherapy Antibodies Resources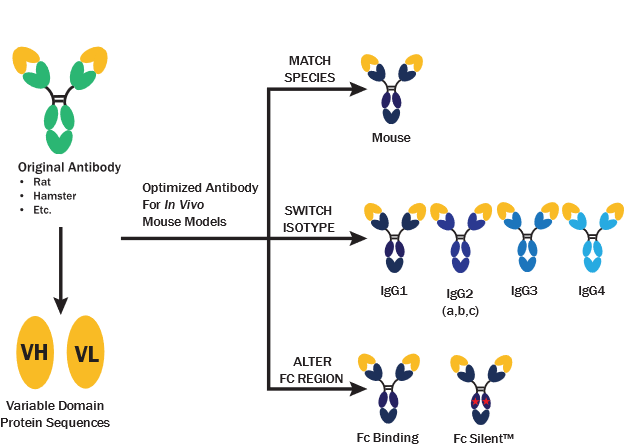
Below find more information on how our engineered recombinant antibodies enable better immunotherapy research, including the potential uses and cited applications for VivopureX™ antibodies against a variety of clinically relevant cancer research targets.
White Paper
A Question of Isotype: How Switching Antibody Isotypes and Fc Regions Promotes Discovery
How do you choose the right antibody format for your project beyond simply matching antibody and antigen? This white paper provides a discussion of how to select the right antibody format for your cancer research in live mouse models.
Case Studies
PD-1 Fc Silent™ Antibody Improves Anti-Tumor Activity in Mice
Researchers at UC Louvain developed a monoclonal antibody blocking GARP:TGF-β1, demonstrating promise in inducing anti-tumor activity. Combining this antibody with the Fc Silent™ anti-PD-1 antibody enhances tumor rejection in mouse models, showcasing a proof-of-concept for potential clinical applications and prompting a first-in-human phase I clinical trial.
Using Bispecific Antibody Reagents to Further Immunotherapy Research
Leiden University Medical Center (LUMC) researchers investigated oncolytic viruses’ synergy with bispecific T-cell-engaging antibodies created in collaboration with our engineers to enhance immunotherapy for solid tumors, demonstrating significant tumor regression and prolonged survival in mouse models.
Research Posters
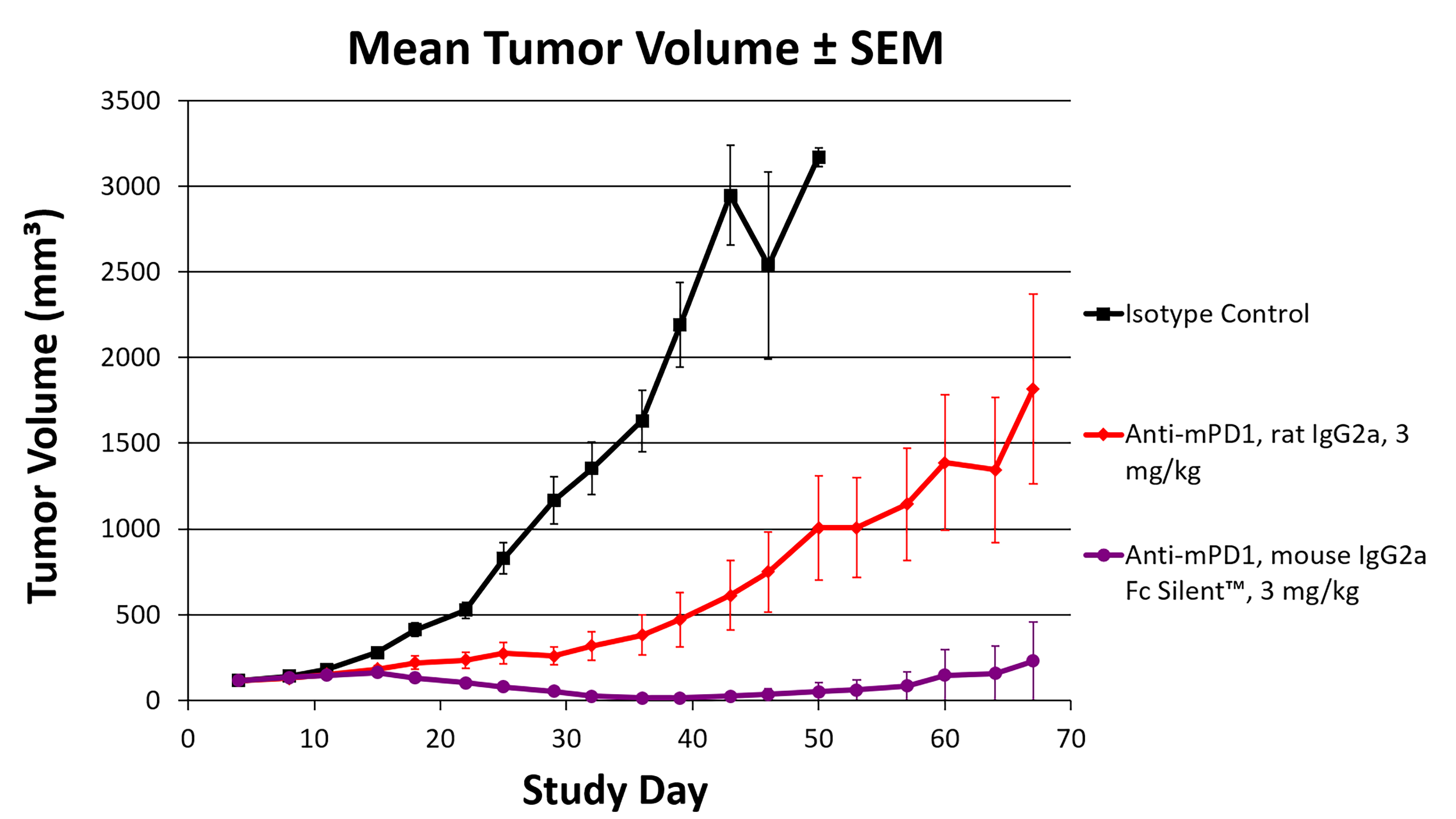 Consistent, long-term PD1 blockade using a syngeneic, engineered anti-PD1 antibody
Consistent, long-term PD1 blockade using a syngeneic, engineered anti-PD1 antibody
Our case study compared VivopureX™ syngeneic mouse IgG2a Fc Silent™ anti-mouse PD-1 antibodies to the original rat IgG2a version. Our engineered VivopureX™ antibody showed better dose efficacy and more homogenous treatment responses, reducing tumor size in mouse models more effectively than the traditional rat monoclonal antibody.
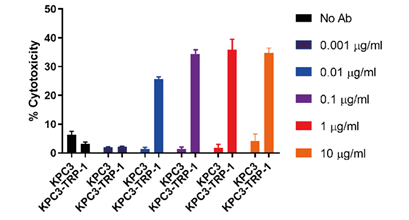 Fully murine knob-into-hole bispecific as surrogate molecule for drug development models
Fully murine knob-into-hole bispecific as surrogate molecule for drug development models
This poster reports the generation of a fully murine, knob-into-hole (KIH), heavy-chain heterodimerizing, bispecific antibody format—the first commercially available production platform of its kind. Additionally, it characterizes an anti-mCD3ε:TRP-1 bispecific antibody, capable of selectively recruiting T-cells to TRP-1+ cancer cells for increased cytotoxic effector function.
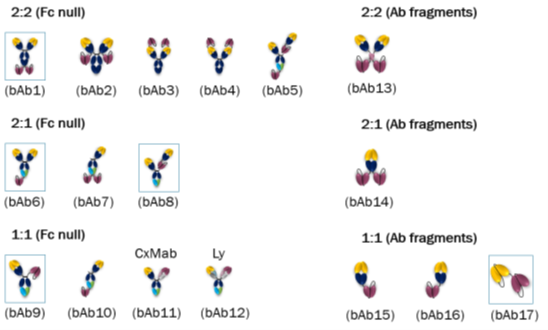 Comparing potential bispecific formats comprising of trastuzumab and a humanized OKT3
Comparing potential bispecific formats comprising of trastuzumab and a humanized OKT3
At Absolute Antibody, we have produced more than 180 different engineered antibody formats. In this case study, we have taken two well-studied monoclonal antibodies and we have shown that not all bispecific formats are created equal.
Product Features
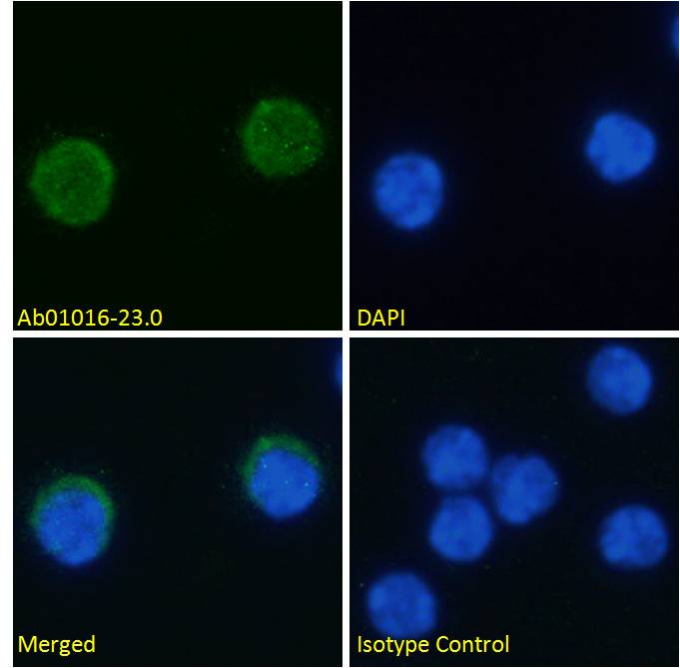
Anti-PD-1H (MH5A)
Immunofluorescence staining of fixed mouse splenocytes with anti-PD-1H antibody MH5A (Ab01016). This antibody is available in a variety of formats, including multiple mouse IgG subtypes and rabbit IgG.
Anti-CTLA-4 (9D9)
Immunofluorescence staining of mouse splenocytes using anti-CTLA-4 antibody (Ab01018) 9D9. This antibody is available in rabbit IgG and a variety of mouse formats, including multiple IgG subtypes, Fab fragments, and bispecific formulations.
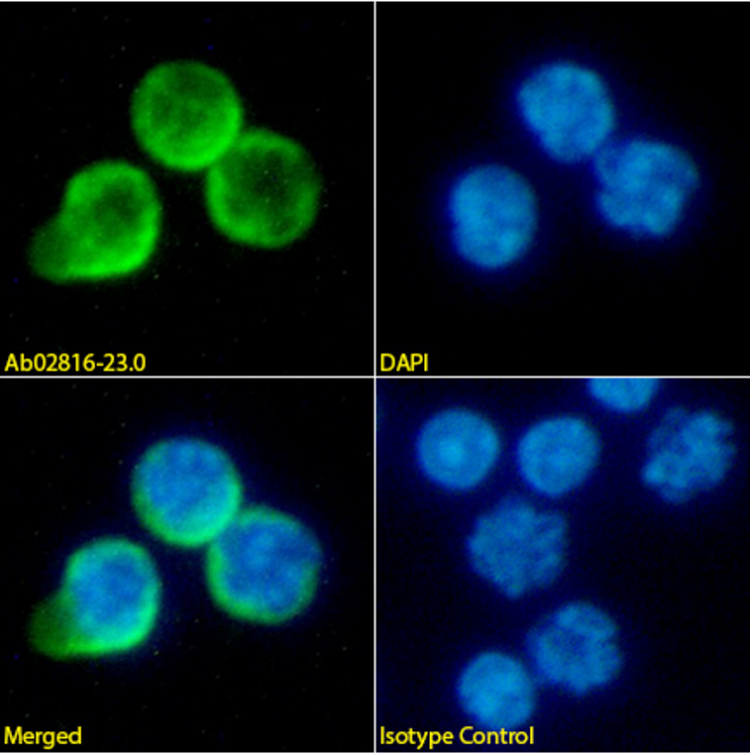
Anti-PD-1 (29F.1A12)
Immunofluorescence staining of mouse splenocytes with anti-PD-1 (Ab02816) 29F.1A12. This antibody is available in multiple mouse IgG subtypes, rat IgG2a and rabbit IgG. Find also antibodies with Fc Silent™ engineering.
- PD-1: The target of several commercially available therapies either approved or under review, the anti PD-1 antibody is a promising tool for cancer research and therapeutic development.
- PD-L1 and PD-L2: This article reviews the therapeutic potential of antibodies that target programmed death ligands, like PD-L1 inhibitors.
- VISTA: Also known as PD-1H, VISTA is another immune checkpoint inhibitor of interest for cancer immunotherapy. Our article reviews the experimental potential of our engineered anti-VISTA antibodies.
- TIGIT: Many tumors use the TIGIT checkpoint to evade anti-cancer immune responses. Our TIGIT antibody engineering expands the study of this pathway with new research potential.
- CD47: Our antibody engineers have created a range of engineered recombinant antibodies targeting CD47, helping researchers elucidate how cancer cells exploit immune checkpoints and new therapeutic targets.
Other Immunotherapy Research Products
As an Absolute Biotech company, we are proud to combine our recombinant portfolio with our sister brands to expand your research possibilities with a wider array of antibodies and reagents.
- Read our article on how to choose the right cancer cell line for your in vivo mouse experiments, with top cell lines like MC-38 and OSCC available through Kerafast.
- Benefit from high-quality antibodies for use in human tissues, like the TRBC1 Mouse anti-Human antibody and KRAS Mouse anti-Human antibody available through LSBio.
- Target proteins at the cutting edge, like the versatile TP53 Tumor Suppressor Protein and the neu-oncogene (HER2) protein complicit in many breast cancers with antibodies from Nordic/Exalpha.
- Explore ELISA kits to streamline your experiments, like the Rat IFNA / Interferon Alpha ELISA Kit and Dog IFN Beta / Interferon Beta ELISA Kit from LSBio.
The critical role of immunotherapy research antibodies in advancing cancer treatments is clear: the right tools can open a wide range of experimental possibilities. With technology like Fc Silent™ mutations to abrogate Fc region binding, species swapping for optimized in vivo performance, and the generation of bispecific antibodies, the wealth of problem-solving capabilities offered by recombinant antibodies is endless. By providing researchers with sophisticated tools like these, the field continues to make strides towards more effective and personalized immunotherapeutic solutions. If your unique research needs have yet to be met by the off-the-shelf antibodies that we have available, we offer custom services such as antibody engineering. For inquiries or quotes, please reach out. Our team of antibody experts is happy to help you.
Immunotherapy Research Antibodies
| Catalog ID | Mouse Target | Antibody Clone |
|---|---|---|
| Ab00110 | CD134 | OX86 |
| Ab00111 | CD200R | OX131 |
| Ab00166 | CD8 alpha/Lyt-2 | YTS 169.4 |
| Ab00205 | CD4 epitope A | YTS 177.9 |
| Ab00206 | CD4 epitope A | YTS 191.1 |
| Ab00295 | Ly6G | 1A8 |
| Ab00584 | CD22 | OX-97 |
| Ab00813 | PD-1 | RMP1-14 |
| Ab00883 | PD-L2 | TY25 |
| Ab00895 | CD27 | RM27-3E5 |
| Ab01017 | PD-1H | mam82 |
| Ab01018 | CTLA-4 | 9D9 |
| Ab01026 | HVEM | HMHV-1B18 |
| Ab01027 | BTLA | HMBT-6B2 |
| Ab01028 | CD80 | RM80 |
| Ab01030 | Ly6G/Ly6C | RB6-8C5 |
| Ab01031 | CD45R | RA3-6B2 |
| Ab01032 | Tim-1 | 3B3 |
| Ab01057 | Tim-3 | 2C12 |
| Ab01058 | Tim-4 | 5G3 |
| Ab01059 | Tim-4 | 3A1 |
| Ab01060 | GITR | DTA-1 |
| Ab01088 | Galectin 9 | RG9-35 |
| Ab01089 | CD153 | RM153 |
| Ab01092 | TIM-2 | RMT2-14 |
| Ab01107 | CD25 | PC-61.5.3 |
| Ab01108 | IFN-gamma receptor 1 | GR20 |
| Ab01110 | CD20 | 18B12 |
| Ab01258 | TIGIT | 1B4 |
| Ab01295 | LAG-3 | C9B7W |
| Ab01297 | CD96 | 6A6 |
| Ab01298 | CD155 | 3F1 |
| Ab01366 | CD226 | 10E5 |
| Ab01417 | PD-1 | J43 |
| Ab01419 | PD-L1 | 10F.9G2 |
| Ab01420 | VISTA | 13F3 |
| Ab01489 | CD19 | 6D5 |
| Ab01490 | IL-10R | 1B1.3a |
| Ab01491 | CD115/M-CSFR | AFS98 |
| Ab01569 | CD47 | mIAP301 |
| Ab02816 | PD-1 | 29F.1A12 |
| Ab00105 | CD3 epsilon | 145-2C11 |
| Ab00109 | Thy1.1 | OX7 |
| Ab00147 | IL-4 | 11B11 |
| Ab00208 | CD5 | YTS 121.5.2 |
| Ab00212 | Thy-1 | YTS 154.7.7.10 |
| Ab00236 | Complement Receptor 1 & 2 | 7G6 |
| Ab00278 | Nerve Growth Factor | alphaD11 |
| Ab00620 | CD62L | MEL-14 |
| Ab00621 | MAdCAM-1 | MECA-367 |
| Ab00449 | alpha 5 beta 1/α5β1 Integrin | M200 (Volociximab) |
| Ab00296 | C5 | Eculizumab |
| Ab00281 | CEA | Arcitumomab |
| Ab04187 | CCL11 | CAT-213 (Bertilimumab) |
| Ab04182 | CCR2 | MLN1202 (Plozalizumab, hu1D9) |
| Ab00726 | CCR4 | KW-0761 (Mogamulizumab) |
| Ab04183 | CD105 | TRC105 (Carotuximab) |
| Ab00535 | CD11a | hu1124 (Efalizumab) |
| Ab01678 | CD154 | IDEC-131 (Toralizumab) |
| Ab00126 | CD20 | 10F381 (rituximab) |
| Ab00728 | CD22 | hL22 (Epratuzumab) |
| Ab00194 | CD25 | YTH 906.9HL (Campath-6) |
| Ab00958 | CD257 | BAFF (Tabalumab) |
| Ab00122 | CD3 epsilon | OKT-3 (muromonab) |
| Ab00283 | CD33 | hP67.6 (Gemtuzumab) |
| Ab00730 | CD33 | SGN-33 (Lintuzumab) |
| Ab00450 | CD4 | CE9.1 (Clenoliximab) |
| Ab00217 | CD4 | MT310 |
| Ab00167 | CD4 | YNB46.1.8 (Campath-9H) |
| Ab00447 | CD40L | hu5c8 (Ruplizumab) |
| Ab00282 | CD41 | 7E3 (Abciximab) |
| Ab04186 | CD49B | GBR500 (Vatelizumab, TMC-2206) |
| Ab00124 | CD52 | Alemtuzumab (Campath-1H) |
| Ab00165 | CD52 | YTH 34.5-G2b (Campath-1G) |
| Ab00736 | CD80 | IDEC-114 (Galiximab) |
| Ab00898 | CD98 | EP3-1 |
| Ab00740 | DR5 | PRO95780 (Drozitumab) |
| Ab01677 | EGFR | Zalutumumab; HuMax-EGFR |
| Ab00279 | EGFR | C225 (Cetuximab) |
| Ab00534 | EGFR | Matuzumab |
| Ab00723 | EGFR domain III | h-R3 (Nimotuzumab) |
| Ab00103 | erbB-2 (Her-2/neu) | 4D5-8 (trastuzumab) |
| Ab04184 | FAP alpha | BIBH1 (Sibrotuzumab, huF19) |
| Ab00802 | Fas | R-125224 |
| Ab03043 | FOLR1 | Farletuzumab (MORAb-003; M3) |
| Ab00717 | IgE | huMaE11 (Omalizumab) |
| Ab00722 | IL-12/23 | ABT-874 (Briakinumab) |
| Ab03711 | IL-20 | h15D2 (Fletikumab) |
| Ab00187 | IL-2R | Daclizumab |
| Ab00188 | IL-2R alpha | Basiliximab |
| Ab04155 | IL-5 | SB-240563 (Mepolizumab) |
| Ab04156 | IL-5 | Sch 55700 (Reslizumab) |
| Ab00737 | IL-6 receptor | rhPM-1 (Tocilizumab) |
| Ab00716 | Integrin alpha 4 | Natalizumab |
| Ab04181 | Integrin alpha-4/beta-7 | Vedolizumab (LDP-02) |
| Ab00773 | Lymphotoxin alpha | MLTA3698A (Pateclizumab) |
| Ab00536 | OX40L | R4930 (Oxelumab) |
| Ab01274 | oxLDL | 2-DO3 |
| Ab00791 | PD-1 | 5C4.B8 (Nivolumab) |
| Ab00729 | RSV | RSHZ19 (Felvizumab) |
| Ab01147 | TAG-72 | Minretumomab (CC49 ) |
| Ab00742 | Tenascin | I-81C6 (Neuradiab) |
| Ab00146 | TNF alpha | cA2 (Infliximab) |
| Ab00448 | TNF alpha | CDP 571 (Humicade) |
| Ab00718 | TNF alpha | D2E7 (Adalimumab) |
| Ab00180 | TRP-1, gp75 | TA99 |
| Ab00280 | Tumor associated glycoprotein (TAG) 72 | B72.3 (Satumomab) |
| Ab00846 | V(beta)3 TCR | JOVI.1 |
| Ab00715 | VEGF | Bevacizumab |
| Ab04185 | Amyloid beta A4 protein | LY2062430 (Solanezumab, hu266) |
| Ab01052 | 4-1BB (CD137) | LOB12.3 |
| Ab01054 | 4-1BBL (CD137L) | AT113-2 |
| Ab00881 | 4-1BBL (CD137L) | TKS-1 |
| Ab01027 | BTLA | HMBT-6B2 |
| Ab00670 | CD27 | LG.3A10 |
| Ab00895 | CD27 | RM27-3E5 |
| Ab00286 | CD28 | E18 |
| Ab00285 | CD28 | D665 |
| Ab01109 | CD40 | 45374 |
| Ab01087 | CD40L (CD154) | MR1 |
| Ab00816 | CD70 | TAN 1-7 |
| Ab01056 | CD70 | TAN1-6 |
| Ab01028 | CD80 (B7-1) | RM80 |
| Ab01029 | CD86 (B7-2) | PO.3 |
| Ab01089 | CD153 | RM153 |
| Ab01297 | CD96 | 6A6 |
| Ab00894 | CTLA-4 | 9H10 |
| Ab01018 | CTLA-4 | 9D9 |
| Ab01088 | GAL-9 | RG9-35 |
| Ab01060 | GITR | DTA-1 |
| Ab00374 | GITR | YGITR 860.103.5 |
| Ab00631 | GITR | YGITR765 |
| Ab00630 | GITRL | YGL386 |
| Ab01026 | HVEM | HMHV-1B18 |
| Ab00814 | ICOS | 7E.17G9 |
| Ab00882 | ICOSL | HK5.3 |
| Ab01295 | LAG3 | C9B7W |
| Ab00110 | OX40 (CD134) | OX86 |
| Ab00564 | OX40L | OX-89 |
| Ab00813 | PD-1 | RMP1-14 |
| Ab01017 | PD-1H (VISTA) | mam82 |
| Ab01016 | PD-1H (VISTA) | MH5A |
| Ab00373 | PD-L1 | YDC 127.1.1 |
| Ab00883 | PD-L2 | TY25 |
| Ab00875 | PDPN (MAP-Tag) | PMab-1 |
| Ab00973 | TIGIT | 4D4 |
| Ab01258 | TIGIT | 1B4 |
| Ab01032 | TIM-1 | 3B3 |
| Ab01110 | TIM-2 | RMT2-14 |
| Ab01057 | TIM-3 | 2C12 |
| Ab01058 | TIM-4 | 5G3 |
| Ab01059 | TIM-4 | 3A1 |
| bAb0249 | CD19xCD3e | 6D5 x 145-2C11 |
| bAb0185 | CD20xCD3e | 18B12 x 145-2C11 |
| bAb0166 | CTLA-4xPD-L1 | 9D9 x VHH-PD-L1 |
| Ab01018-1.169 | CTLA-4xOX40 | 9D9 x OX86 |
| bAb0163 | EGFRxCD3e | C225 (Cetuximab) x 145-2C11 |
| bAb0164 | EGFRvIIIxCD3e | L8A4 x 145-2C11 |
| bAb0162 | erbB-2 (Her-2/neu)xCD3e | 4D5-8 (trastuzumab) x 145-2C11 |
| bAb0183 | erbB-2 (Her-2/neu)xCD3e | 4D5-8 (trastuzumab) x OKT3 |
| bAb0184 | F4/80xCD3e | recombinant Cl:A3-1 x 145-2C11 |
| bAb0161 | FluoresceinxCD3e | 4-4-20 (enhanced) x 145-2C11 |
| bAb0168 | FluoresceinxPD-L1 | 4-4-20 (enhanced) x VHH-PD-L1 |
| bAb0167 | FluoresceinxCD47 | 4-4-20 (enhanced) x A4 |
| bAb0136 | TRP-1, gp75 x CD3e | TA99 x 145-2C11 |
| bAb0165 | TRP-1, gp75 x CD47 | TA99 x A4 |
| bAb0182 | TRP-1, gp75 x OX40 | TA99 x OX86 |
| Ab00542-1.68 | CD200 x CD47 | OX-90 |
| Ab00813-1.69 | PD-1 x PD-L1 | RMP1-14 |
| Ab00813-1.68 | PD-1 x CD47 | RMP1-15 |
| bAb0615 | PD-L1 x CD3e | 10F.9G2 x 145-2C11 |
| Ab00373-1.68 | PD-L1 x CD47 | YDC 127.1.1 |
| Ab01419-1.169 | PD-L1 x OX40 | 10F.9G2 x OX86 |
| bAb0653 | PD-L1 x CD28 | 10F.9G2 x D665 |
| Ab01017-1.69 | PD-1H x PD-L1 | mam82 |
| Ab00973-1.69 | TIGIT x PD-L1 | 4D4 |
| bAb0505 | NKG2D x CD3e | CX-5 x 145-2C11 |
| Ab00782 | A33 | huA33 |
| Ab00213 | Alpha-fetoprotein (AFP) | AF5 |
| Ab02189 | Alpha-ketoglutarate-dependent dioxygenase alkB homolog 3 | 32C8_71 |
| Ab03472 | Aspartate beta-hydroxylase | G3-F11 |
| Ab01321 | BCL2 | bcl-2/100 |
| Ab00181 | Beta-2 microglobulin | BBM.1 |
| Ab01322 | BLC2 | bcl-2/124 |
| Ab00380 | Breast Epithelial Mucin | BrE-3 |
| Ab01215 | c-erbB-2 | FWP51 |
| Ab00525 | CA125 | 196-14 |
| Ab02836 | CA125 | OC125-3.11 |
| Ab02835 | CA125 | VK-8-1.9 |
| Ab03769 | CA19-9 | 4A6 |
| Ab03770 | CA19-9 | 4G2 |
| Ab03766 | CA242 | C242 |
| Ab03768 | CA50 | CA50Ab01 |
| Ab02092 | Canine Lymphoma | Mab231 |
| Ab00281 | carcinoembryonic antigen (CEA) | Arcitumomab |
| Ab01165 | carcinoembryonic antigen | A5B7 |
| Ab03901 | CD155 | CD155.16 |
| Ab03902 | CD155 | scFv1 |
| Ab03903 | CD155 | scFv3 |
| Ab03739 | CD155 | SKII.4 |
| Ab02210 | CD19 | B4 |
| Ab02064 | CEA | BW431/26 |
| Ab02721 | CEA | CH1A1A (2F1, CH1A1A-2F1) |
| Ab01002 | CEA | EB-011 |
| Ab01003 | CEA | EB-022 |
| Ab02833 | CEA | T84.66 |
| Ab02587 | CIP2A | HL1925 |
| Ab01683 | CSAp | Mu-9 |
| Ab00616 | Ctip2 | 25B6 |
| Ab01648 | Cytokeratin 7 | OV-TL 12/30 |
| Ab03179 | Cytokeratin 8 | TS1 |
| Ab00822 | deltaEGFR | DH8.3 |
| Ab01677 | EGFR | 2F8 (Zalutumumab; HuMax-EGFR) |
| Ab02461 | EGFR | 2224 |
| Ab00133 | EGFR | 528 |
| Ab00279 | EGFR | C225 (Cetuximab) |
| Ab03275 | EGFR | cet.Hum (Cetuximab – humanized; hC225) |
| Ab00608 | EGFR | EGFR1 |
| Ab00453 | EGFR | L2 1C |
| Ab00455 | EGFR | L3 11D |
| Ab00534 | EGFR | Matuzumab |
| Ab00452 | EGFR | S3 12D |
| Ab00454 | EGFR | S4 10H |
| Ab00723 | EGFR domain III | h-R3 (Nimotuzumab) |
| Ab00184 | EGFRvIII | L8A4 |
| Ab03195 | Endosialin | scFv78 |
| Ab01197 | EPCAM | B38.1 |
| Ab00103 | erbB-2 (Her-2/neu) | 4D5-8 (trastuzumab) |
| Ab00303 | ERBB2 | SAIC-02A-7 |
| Ab00515 | GA733-2 | FU-MK-1 |
| Ab03700 | GDF15 | Clone 29 |
| Ab03701 | GDF15 | MBM-12 |
| Ab03702 | GDF15 | MBM-14 |
| Ab03427 | Glypican-1 | AbAb-GPC1 |
| Ab01093 | HER2 | 4D5 |
| Ab00924 | HER2 | C7b |
| Ab02462 | HER2 | ML3-9 (C6ML3-9) |
| Ab00383 | HER2/neu | SER4 |
| Ab02460 | HER3 | A5 |
| Ab02661 | HER4 | B6 (HER4.B6) |
| Ab01485 | high MWT epithelial antigens | KC4G3 |
| Ab00430 | Human Ephrin Type A receptor 2 | 1C1 |
| Ab03260 | JAM-A | 6F4 |
| Ab03878 | KRAS G12C mutant | AbAbKRAS01 |
| Ab03877 | KRAS G12D mutant | D113 |
| Ab01227 | Lewis Y | 3S193 |
| Ab00492 | LeY | BR96 |
| Ab00493 | LeY | H18A |
| Ab00761 | LMP1 | H3 |
| Ab01565 | m5C | FMC-9 |
| Ab02838 | MAGEA3 | 21B4 |
| Ab02080 | Mesothelin | 11-25 |
| Ab02078 | Mesothelin | C10 |
| Ab02081 | Mesothelin | IC14-30 |
| Ab02079 | Mesothelin | K1 |
| Ab02082 | MPF | 20-10 |
| Ab02083 | MPF | 41-28 |
| Ab01329 | MUC1 | E29 |
| Ab02206 | MUC1 | HMFG1 (1.10.F3) |
| Ab00712 | MUC1 | HMFG2 |
| Ab00345 | MUC1 | Mc5 |
| Ab02454 | MUC1 | PAM4 |
| Ab00215 | MUC1 | SM3 |
| Ab03096 | NSE | E1-7E2 |
| Ab03097 | NSE | E2-4F6 |
| Ab00512 | OA3 | OV-TL3 |
| Ab01168 | osteosarcoma-associated cell membrane antigen | TP-1 |
| Ab01167 | osteosarcoma-associated cell membrane antigen | TP-3 |
| Ab00923 | p21 | Y13-238 |
| Ab03876 | p21Ras | DWP |
| Ab03875 | p21Ras | KGH-R1 |
| Ab00142 | p53 | PAb421 |
| Ab01166 | polymorphic epithelial mucin | CTM01 |
| Ab00423 | PSA | 8G8F5 |
| Ab00435 | PSA | scFv B8 |
| Ab03135 | PSMA | 11d1e5E10 |
| Ab03969 | PSMA | 8C12 |
| Ab03136 | PSMA | J591 |
| Ab01325 | S100 | S1-61 |
| Ab02453 | S100B | 1C8 |
| Ab00301 | Synaptophysin | SP15 |
| Ab01147 | TAG-72 | Minretumomab (CC49) |
| Ab00742 | Tenascin | I-81C6 (Neuradiab) |
| Ab00382 | Tn Antigen | 237mAb |
| Ab00619 | trastuzumab (anti-idiotype) | scFv40 |
| Ab00618 | trastuzumab (anti-idiotype) | scFv69 |
| Ab00180 | TRP-1, gp75 | TA99 |
| Ab00280 | Tumor associated glycoprotein (TAG) 72 | B72.3 (Satumomab) |
| Ab00169 | VAP-1 | 2D10 |
| Ab02395 | VAP-1 | 3F10 |
| Ab02394 | VAP-1 | 3G6 |
| Ab00168 | VAP-1 | TK8-14 |

 United States (US)
United States (US)